We design materials with theories, computations and data.
|
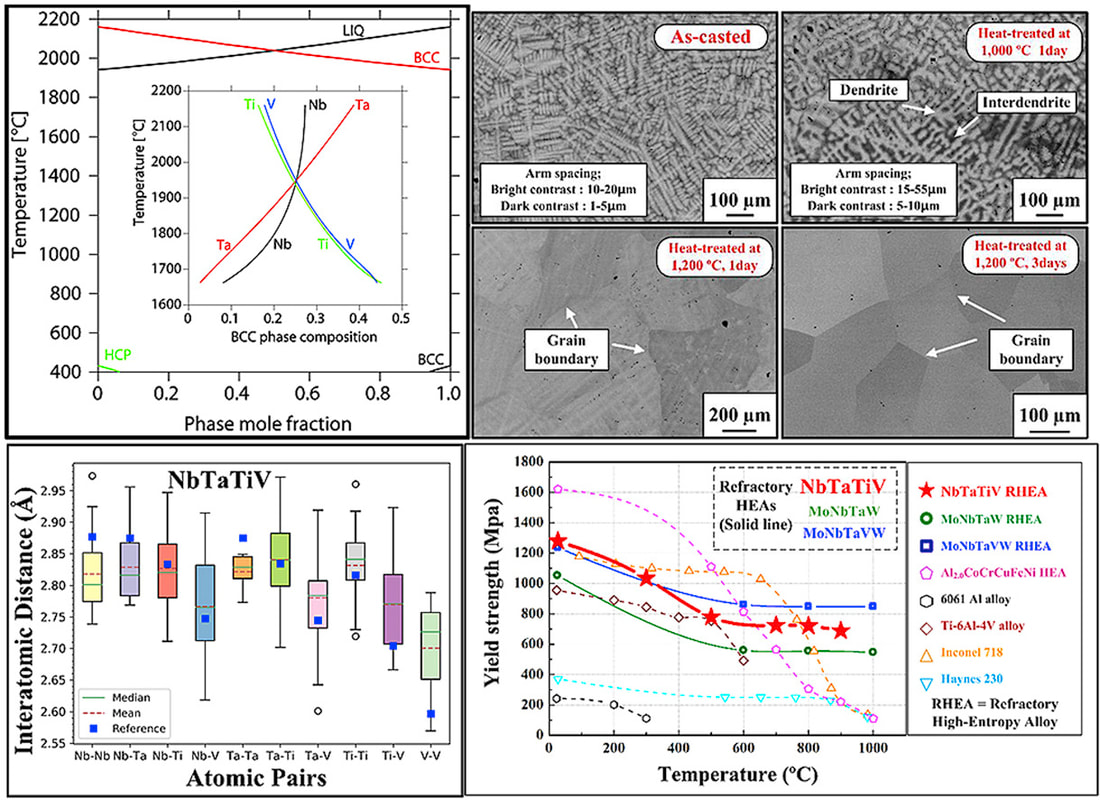
In collaboration with Prof. Peter Liaw's group at University of Tennessee, we recently designed a new single-phase BCC refractory high-entropy alloy (HEA) NbTaTiV. The novel HEA exhibits high yield strength and ductility at both room and high temperatures. First-principles approaches based on density functional theory (DFT) provided critical information on the thermodynamic stability and lattice distortions in the alloy design. Integrated with the CALPHAD method and in situ structural characterizations, a heat-treatment process was developed that eliminates structural and chemical inhomogeneity. "Lattice distortion in a strong and ductile refractory high-entropy alloy", Acta Materialia, 160, 158-172 (2018) [PDF]
0 Comments
Refractory high-entropy alloys (RHEA) are promising structural materials for high-temperature thermal-harsh environment. The HfNbTaTiZr RHEA is a good example that shows a rare combination of high strength and good ductility. In real-world applications, high-temperature alloys have to maintain a high phase stability not only at high temperatures, but also for a wide range of temperatures for a prolonged service time. While the HfNbTaTiZr RHEA is generally regarded as a single-phase BCC solid solution, recent studies by Senkov et al suggest phase decomposition after cold-rolling at 800 °C. Controversy still exists for the phase stability of the HfNbTaTiZr RHEA at intermediate temperatures. In the present work, we investigated the phase decomposition of the RHEA at different temperatures (500–1000 °C). The formation of BCC Ta-Nb-rich and HCP HfZr-rich precipitates, as well as their preferred orientation to the BCC matrix, are elucidated from experiments. Thermodynamic modeling shows good agreement with experiments.
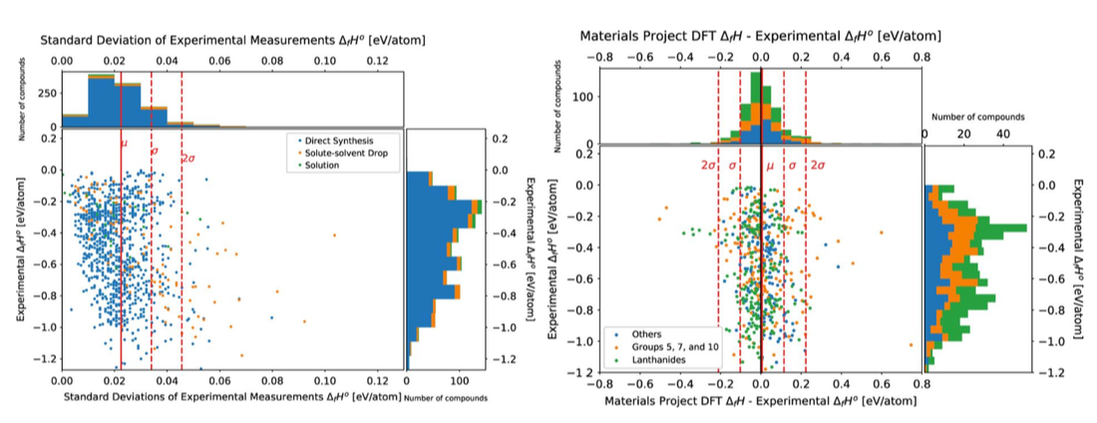
"Phase transformations of HfNbTaTiZr high-entropy alloy at intermediate temperatures", Scripta Materialia, 158, 50-56 (2019) [PDF] An open database of experimental formation enthalpy data for > 1200 metallic phases released10/18/2017 The standard enthalpy of formation is a fundamental thermodynamic property that determines the phase stability of a compound, which can be coupled with other thermodynamic data to calculate phase diagrams. Calorimetry provides the only direct method by which the standard enthalpy of formation is experimentally measured. However, the measurement is often a time and energy intensive process. We present a dataset of enthalpies of formation measured by high-temperature calorimetry. The phases measured in this dataset include intermetallic compounds with transition metal and rare-earth elements, metal borides, metal carbides, and metallic silicides. The dataset contains 1,276 entries on experimental enthalpy of formation values and structural information. Most of the entries are for binary compounds but ternary and quaternary compounds are being added as they become available.
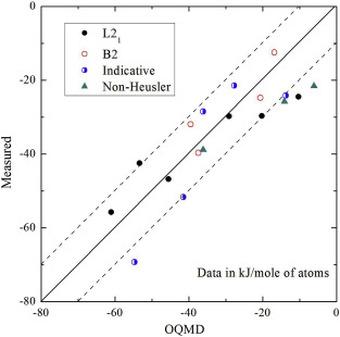
The dataset also contains predictions of enthalpy of formation from first-principles calculations for comparison. We compared DFT formation enthalpy values from the Materials Project and OQMD, and identified problematic systems that show substantial discrepancies between experiments and PBE-DFT. The most recent database can be queried from our website: http://tptc.iit.edu/ The data file can be accessed from Figshare: https://doi.org/10.6084/m9.figshare.c.3822835 Experimental formation enthalpies for intermetallic phases and other inorganic compounds", Scientific Data, 4, 170162 (2017) [PDF] We recently collaborated with Prof. Philip Nash's group to investigate the formation enthalpies of a series of Ni2YZ Heusler compounds. This paper includes one of the most comprehensive experimental formation enthalpy data of the Ni2YZ Heusler family. The experimental data offer a rare opportunity to evaluate the DFT accuracy for these important compounds.
Please check out Prof Nash's website, which gives open access to a database of experimental formation enthalpies for tens of hundreds of compounds. As another strong testimony of the power of high-throughput calculations, the Materials Project just released the world's largest database of piezoelectric properties for hundreds of compounds. Our paper on Scientific Data describes the details of the dataset, but the importance of the data is far beyond the technical details: We might be able to discover materials with outstanding piezoelectric properties from this growing database.
See the report from the Berkeley Lab News Center: ✐ Computing a textbook of crystal physics: Berkeley lab scientists publish world's largest database of piezoelectric properties, Berkeley Lab News Center Please also check out our earlier paper on the world's largest database of elastic tensors: ✐ Accelerating materials discovery with world's largest database of elastic properties: Berkeley lab scientists increase by almost ten-fold the data previously available |
Categories
All
Archives
September 2020
|


 RSS Feed
RSS Feed
