We design materials with theories, computations and data.
|
Glad to be a member of a team that received the 2017 National Energy Research Scientific Computing Center (NERSC) Award for High-Impact Scientific Achievement:
A team of researchers from Lawrence Berkeley National Laboratory, University of California Berkeley and Caltech was honored in this category for using NERSC resources to speed up the discovery of commercially viable materials that can be used to produce solar fuels. The group gathered a list of potentially useful compounds and then were able to rapidly screen and test the best materials with NERSC. This process would normally take an immense amount of time to conduct all the tests and experiments by hand. The researchers were able to go through 174 compounds containing vanadium and oxygen, called vanadates, and were able to identify 12 useful materials. These materials will be very useful for developing solar fuels, which are a clean and renewable alternative to fossil fuels. The work was led by Berkeley Lab's Jeff Neaton, John Gregoire and Qimin Yan. Other members of the team were Jie Yu, Santosh Suram, Lan Zhou, Aniketa Shinde, Paul Newhouse, Wei Chen, Guo Li and Kristin A. Persson.
1 Comment
We are glad to receive a 3-year award from the Solid State & Materials Chemistry Program of National Science Foundation (DMR-1709959) to study mechanical activation enhanced solid-state reaction and electrochemical properties of the NaCrO2 cathode. The work will be in collaboration with Prof. Leon Shaw.
Dr. Chen received a 3-year award from the Metals & Metallic Nanostructure program of National Science Foundation (DMR-1607943) to study the thermodynamic properties of binary and ternary intermetallic compounds using experimental and computational methods. This work will be in collaboration with Prof. Philip Nash.
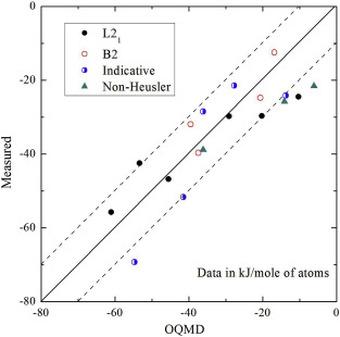
We recently collaborated with Prof. Philip Nash's group to investigate the formation enthalpies of a series of Ni2YZ Heusler compounds. This paper includes one of the most comprehensive experimental formation enthalpy data of the Ni2YZ Heusler family. The experimental data offer a rare opportunity to evaluate the DFT accuracy for these important compounds.
Please check out Prof Nash's website, which gives open access to a database of experimental formation enthalpies for tens of hundreds of compounds. As another strong testimony of the power of high-throughput calculations, the Materials Project just released the world's largest database of piezoelectric properties for hundreds of compounds. Our paper on Scientific Data describes the details of the dataset, but the importance of the data is far beyond the technical details: We might be able to discover materials with outstanding piezoelectric properties from this growing database.
See the report from the Berkeley Lab News Center: ✐ Computing a textbook of crystal physics: Berkeley lab scientists publish world's largest database of piezoelectric properties, Berkeley Lab News Center Please also check out our earlier paper on the world's largest database of elastic tensors: ✐ Accelerating materials discovery with world's largest database of elastic properties: Berkeley lab scientists increase by almost ten-fold the data previously available After three years in California, I moved back to Chicago and joined Illinois Institute of Technology as an assistant professor in the Department of Mechanical, Materials & Aerospace Engineering.
|
Categories
All
Archives
September 2020
|

 RSS Feed
RSS Feed
